Action required: Please refresh your browser
We have recently implemented some changes that require a hard refresh of your browser: Please hold down the CTRL-key and press the F5 key.
After a successful hard refresh, this message should not appear anymore.
More details about this topic are available here »
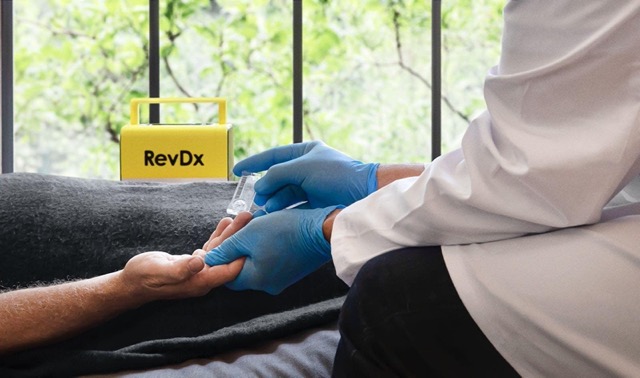
| EFA Technologies, pioneer in portable blood labs, enters Brazilian market after Anvisa approval of its portable RevDx device | ||
| By: GlobeNewswire - 10 Apr 2024 | Back to overview list |
|
| EFA also secures US$6.5 Million in funding, backed by existing investors and new partners Tel Aviv, Israel, 10 April 2024 - EFA Technologies, pioneers in portable blood labs, today announces the start of its operations in Brazil, following approval last October of its leading portable blood lab device, RevDx, by Anvisa, the Brazilian health regulatory authority. The company also secured US$6.5 million in funding, boosting EFA’s mission to further improve healthcare worldwide. RevDx, EFA’s flagship product, is a sophisticated portable diagnostic device that leverages advanced technology to provide accurate and rapid medical testing locally. Its capabilities include identifying pathogens, analyzing biomarkers and aiding in disease diagnosis. Thereby revolutionizing the field of on-site diagnostics with its efficiency and precision. RevDx marks a significant breakthrough in medical diagnostics. It is portable, affordable and user-friendly, leveraging microscopy and AI to deliver instant results. This not only reduces costs and saves time, but also improves the doctor-patient relationship. With the capability to perform a full CBC count from just a single drop of blood, the device streamlines diagnostics and equips healthcare professionals with actionable insights. Yoel Ezra, CEO of EFA Technologies: Backed by a distinguished medical board that includes Prof. Aaron Ciechanover, a 2004 Nobel Prize Laureate, EFA Technologies is poised to revolutionize healthcare on a global scale. Prof. Ciechanover's endorsement underscores the significance of RevDx's medical innovation. EFA Technologies has obtained CE certification and enters the Brazilian market with ANVISA certification for RevDx. Meanwhile, EFA is actively exploring new markets for further expansion. Amir Lubshevsky, EFA’s Chief Medical Officer, adds: New financing round
RevDx, is a sophisticated portable diagnostic device that leverages advanced technology to provide accurate and rapid medical testing locally. Its capabilities include identifying pathogens, analyzing biomarkers and aiding in disease diagnosis. Thereby revolutionizing the field of on-site diagnostics with its efficiency and precision. About EFA Technologies EFA’s business strategy is to develop its RevDx solution as a platform that will enable the creation of an ecosystem that can develop more diagnostic applications over time. RevDx innovation fits this approach in both its design and capabilities. EFA Technologies packages multi-channel capabilities into the device, starting with automatic microscopy and bio-sensing readers. The digital images and data generated from the system are the basis for further image-processed diagnostics and algorithms. As EFA Technologies continues its journey of innovation and expansion, its commitment to improving healthcare accessibility and effectiveness remains steadfast. With RevDx at the forefront and underpinned by robust financial backing, EFA is primed to redefine medical diagnostics and enhance patient outcomes worldwide. For more info please visit: efa-technologies.com, or find us on LinkedIn Our legal advisors: Naschitz Brandes Amir Ltd. www.nblow.com Contact for media: EFA Technologies LifeSpring Life Sciences Communication, Amsterdam, the Netherlands Attachment 
|
||
|
|
||
 | Back to overview list | |